That the Earth has undergone sudden large changes is demonstrated by all manner of geoscientific records. It seems that many of these catastrophic events occurred whenever steady changes reach thresholds that trigger new behaviours in the interlinked atmosphere, hydrosphere, atmosphere, biosphere and lithosphere that constitute the Earth system. The driving forces for change, both steady and chaotic, may be extra-terrestrial, such as the Milankovich cycles and asteroid impacts, due to Earth processes themselves or a mixture of the two. Our home world is and always has been supremely complicated; the more obviously so as knowledge advances. Abrupt transitions in components of the Earth system occur when a critical forcing threshold is passed, creating a ‘tipping point’. Examples in the geologically short term are ice-sheet instability, the drying of the Sahara, collapse of tropical rain forest in the Amazon Basin, but perhaps the most important is the poleward transfer of heat in the North Atlantic Ocean. That is technically known as the Atlantic Meridional Overturning Circulation with the ominous acronym AMOC.

As things stand today, warm Atlantic surface water, made more saline and dense by evaporation in the tropics is transferred northwards by the Gulf Stream. Its cooling at high latitudes further increases the density of this water, so at low temperatures it sinks to flow southwards at depth. This thermohaline circulation continually pulls surface water northwards to create the AMOC, thereby making north-western European winters a lot warmer than they would be otherwise. Data from Greenland ice cores show that during the climatic downturn to the last glacial maximum, the cooling trend was repeatedly interrupted by sudden warming-cooling episodes, known as Dansgaard-Oeschger events, one aspect of which was the launching of “armadas” of icebergs to latitudes as far south as Portugal (known as Heinrich events), which left their mark as occasional gravel layers in the otherwise muddy sediments on the deep Atlantic floor (see: Review of thermohaline circulation; February 2002).
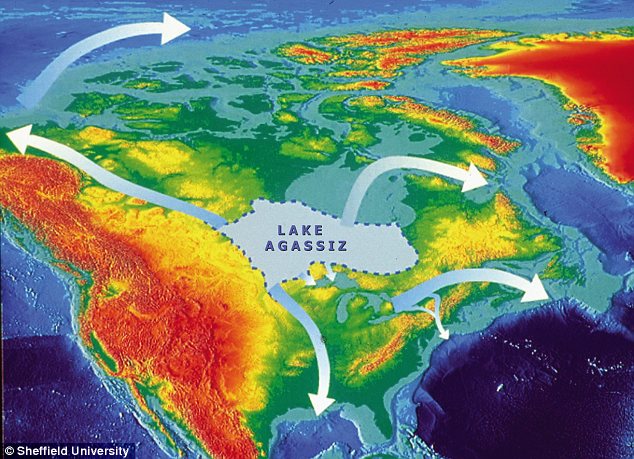
These episodes involved temperature changes over the Greenland icecap of as much as 15°C. They began with warming on this scale within a matter of decades followed by slow cooling to minimal temperatures, before the next turn-over. Various lines of evidence suggest that these events were accompanied by shutdowns of AMOC and hence the Gulf Stream, as shown by variations in the foraminifera species in sea-floor sediments. The culprit was vast amounts of fresh water pouring into the Arctic and northernmost Atlantic Oceans, decreasing the salinity and density of the surface ocean water. In these cases that may have been connected to repeated collapse of circumpolar ice sheets to launch Heinrich’s iceberg armadas. A similar scenario has been proposed for the millennium-long Younger Dryas cold spell that interrupted the onset of interglacial conditions. In that case the freshening of high-latitude surface water was probably a result of floods released when glacial barriers holding back vast lakes on the Canadian Shield burst.
At present the Greenland icecap is melting rapidly. Rising sea level may undermine the ice sheet’s coastal edges causing it to surge seawards and launch an iceberg armada. This may be critical for AMOC and the continuance of the Gulf Stream, as predicted by modelling: counter-intuitive to the fears of global warming, at least for NW Europe. In August 2024 scientists from Germany and the UK published what amounts to a major caution about attempts to model future catastrophes of this kind (Ben-Yami, M. et al 2024, Uncertainties too large to predict tipping times of major Earth system components from historical data, Science Advances, v. 10, article eadl4841; DOI 10.1126/sciadv.adl4841). They focus on records of the AMOC system, for which an earlier modelling study predicted that a collapse could occur between 2025 and 2095: of more concern than global warming beyond the 1.5° C currently predicted by greenhouse-gas climate models .
Maya Ben-Yami and colleagues point out that the assumptions about mechanisms in Earth-system modelling and possible social actions to mitigate sudden change are simplistic. Moreover, models used for forecasting rely on historical data sets that are sparse and incomplete and depend on proxies for actual variables, such as sea-surface and air temperatures. The further back in geological time, the more limited the data are. The authors assess in detail data sets and modelling algorithms that bear on AMOC. Rather than a chance of AMOC collapse in the 21st century, as suggested by others, Ben Yami et al. reckon that any such event lies between 2055 and 8065 CE, which begs the question, “Is such forecasting worth the effort?”, however appealing it might seem to the academics engaged in climatology. The celebrated British Met Office and other meteorological institutions, use enormous amounts of data, the fastest computers and among the most powerful algorithms on the planet to simulate weather conditions in the very near future. They openly admit a limit on accurate forecasting of no more than 7 day ahead. ‘Weather’ can be regarded as short-term climate change.
It is impossible to stop scientists being curious and playing sophisticated computer games with whatever data they have to hand. Yet, while it is wise to take climate predictions with a pinch of salt because of their gross limitations, the lessons of the geological past do demand attention. AMOC has shut down in the past – the last being during the Younger Dryas – and it will do so again. Greenhouse global warming probably increases the risk of such planetary hiccups, as may other recent anthropogenic changes in the Earth system. The most productive course of action is to reduce and, where possible, reverse those changes. In my honest opinion, our best bet is swiftly to rid ourselves of an economic system that in the couple of centuries since the ‘Industrial Revolution’ has wrought these unnatural distortions.