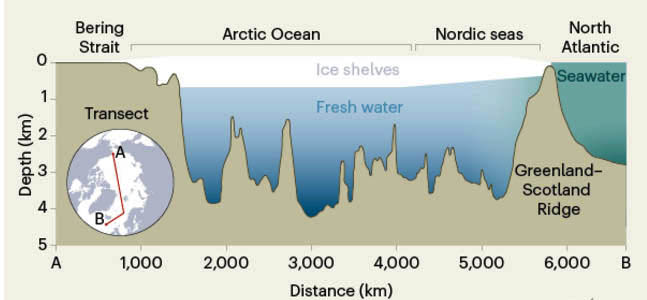
The salinity of surface water at high latitudes in the North Atlantic is a critical factor in its sinking to draw warm, low-latitude water northwards in the Gulf Stream while contributing to the southwards flow of North Atlantic Deep Water along the ocean floor. One widely supported hypothesis for rapid cooling events, such as the Younger Dryas, is the shutdown of this thermohaline circulation (Review of thermohaline circulation, February 2002). That may happen when surface seawater at high latitudes is freshened and made less dense by rapid melting or break-up of continental ice sheets, or through the release of vast amounts of fresh water from glacially dammed lakes. The climatic decline leading to the last glacial maximum at around 20 ka was punctuated by irregular episodes known as Dansgaard-Oeschger and Heinrich Events that have been attributed to such hiccups in thermohaline processes. In this context, a whole new barrel of fish has been opened up by a geochemical study of the top few metres of sediments on the Arctic Ocean floor (Geibert, W. et al. 2021. Glacial episodes of a freshwater Arctic Ocean covered by a thick ice shelf. Nature, v. 590, p. 97–102; DOI: 10.1038/s41586-021-03186-y), particularly their content of an isotope of thorium (230Th).
Being radioactive (half-life ~75 ka), 230Th is useful in working out sediment deposition rates, especially as it is insoluble and adheres to dust grains. The isotope is a decay product of uranium, yet it not only forms on land from uranium in hard rocks, eventually to be transported into marine sediments, but from uranium dissolved in seawater too. Interestingly, the amount of uranium that can enter seawater in solution depends on water salinity. Fresh water, especially that locked up in glacial ice, has very low concentrations of uranium. Consequently, ordinary seawater adds additional 230Th to sediments whereas fresh water does not. An excess of the isotope in marine sediments signifies their deposition from salty water, but those deposited in fresh water carry no excess. In the course of analysing deep-sea cores from the floors of the Arctic Ocean and the northernmost part of the North Atlantic, Walter Geibert and colleagues at the Alfred Wegener Institute in Bremerhaven, and the University of Bremen, Germany revealed a series of sediment layers that were devoid of excess 230Th. This suggests that twice, probably in periods between 150 to 131 and 70 to 62 ka, water in the Arctic Ocean and the connected Nordic Sea was entirely fresh. In two cores the evidence suggests a third, restricted occurrence of fresh water fill at about 15 ka.
The most likely explanation is that the fresh-water episodes marked the development of major ice shelves, similar to those still present around Antarctic; i.e. floating or grounded ice of glacial origin (not sea ice). That had been anticipated, but not previously proved for the northern polar region. The outlets from the Arctic Ocean basin to the Pacific and North Atlantic Oceans are marked by barriers of shallow seabed. One is the Bering Straits, which became the Beringia land bridge that facilitated animal and human migrations from Siberia to North America when sea level fell as continental ice sheets grew. The other is the Greenland-Scotland Ridge formed by volcanism connected to the Icelandic hot spot as the North Atlantic opened. It is possible that the suggested ice shelves grounded on these ridges, to effectively dam and isolate the Arctic Ocean. Fresh water from melting land ice would ‘pond’ beneath the ice shelves, floating on denser salt water and eventually expelling it from much of the polar marine basin. A side effect of this would have been partially to accumulate and isolate the oxygen-isotope proportions that characterise snow and glacial ice. Remember that the light 16O isotope is preferentially extracted from sea water during evaporation, to become stored in glacial ice sheets so that the proportion of the heavier 18O increases in ocean water; δ18O is therefore an important proxy for glacial waxing and waning and thus the fluctuations of global sea level. Trapping a proportion of water of glacial origin in isolated Arctic Ocean water and ice shelves would explain discrepancies in the oxygen-isotope records of successive ice ages. Also, if the ice shelves periodically broke up, fresh water derived from them and ponded in the deepest Arctic Ocean basin could change the salinity of surface ocean water elsewhere – being lower density that fresh water would ‘float’.
The work of Geibert and colleagues may well result in a great deal of head scratching among palaeoclimatologists and perhaps new ideas on the dynamics of ice age climates.
See also: Hoffmann, S. 2021. The Arctic Ocean might have been filled with freshwater during ice ages. Nature, v. 590, p. 37-38; DOI: 10.1038/d41586-021-00208-7