I have been following the harrowing story of how arsenic gets into domestic water supplies for 20 years (see: Earth-logs Geohazards for 2002; 2003; 2004; 2005; 2006; 2008; 2009; 2011; 2013; 2017). In my opinion, it is the greatest natural hazard in terms of the numbers at risk of poisoning. In 2006 I wrote about the emergence in Bangladesh of arsenic poisoning on a huge scale during the mid 1990s for a now defunct Open University course. If people depend for drinking water on groundwater from tube wells driven into alluvium they would not know of the risk, unless the water is rigorously analysed for levels of As greater than 10 micrograms per litre (μg l-1), the WHO recommended maximum. The sad fact is that the affected population were advised to switch from surface water supplies, which carry a high risk of biological infection, to well water. That is because during downward percolation from the surface oxidation destroys bacteria and viruses as well as parasites. Opportunities provided by a massive UN-funded drilling programme and local well digging made the choice seemingly obvious. Most people came to prefer well water as gastro-intestinal infections and child mortality fell rapidly.
Arsenic adds no taste, which is why it was once the ‘poison of choice’. How it gets into groundwater is difficult to judge, unless wells are downflow of areas riddled with metal mines. Years of research uncovered an unsuspected mechanism. The most common colorant of mineral grains, and thus sedimentary rocks, is brownish iron hydroxide (goethite), and that is able to adsorb a range of dissolved elements, including arsenic. One would think, therefore, that groundwater should be made safe by such a natural ‘filtering’ process: indeed goethite can be used in decontamination. The problem is that iron hydroxide, which contains Fe-3, is only stable in water with a high capacity for oxidation. Under reducing conditions it breaks down to soluble Fe-2 and water, thereby releasing to solution any other element that it has adsorbed. In alluvium, beds containing organic matter are prone to this ‘reductive dissolution’ of goethite. If weathering upstream has released even seemingly insignificant amounts of arsenic during the build up of alluvium, there is a potential life-threatening problem as arsenic builds up in the goethite coating of sedimentary grains to become ‘locked in’, with the potential to be released in high concentrations if subsurface chemical conditions change. The colour of the alluvial sediments penetrated by wells is a clue. If they are reddish brown, groundwater is safe, if they are greyish and goethite-free then, ‘beware’. But it is rare to examine ‘cuttings’ from a drill site aimed at groundwater, unlike those aimed at ores or oil
Since the tragedy of Bangladesh, which resulted after 5 years or so in obvious signs of arsenicosis – dark wart-line keratoses on hands and feet or black blotches on facial and torso skin – several alluvial basins in large river systems have had their well water tested. But by no means all such basins have been screened in this way, and there are many less-obvious signs of arsenic poisoning. After long exposure to the lower range of dangerous arsenic levels a variety of cancers develop in known areas of arsenic risk. There are also high levels of endemic respiratory problems, cardiovascular disease, reduced intellectual development in children and even diabetes. Geochemical monitoring of all populated and farmed river systems is a huge task that is far beyond the resources of many countries through which they run. One approach to ‘screening’ for hazard or safety is to use geological, hydrological, soil, climate and topographic data. Those from known arsenic-prone basins and those where its levels are shown to be consistently below the 10 μg l-1 danger threshold help to develop a predictive model (Podgorski, J. & Berg, M. 2020. Global threat of arsenic in groundwater. Science, v. 368, p. 845-850; DOI: 10.1126/science.aba1510).
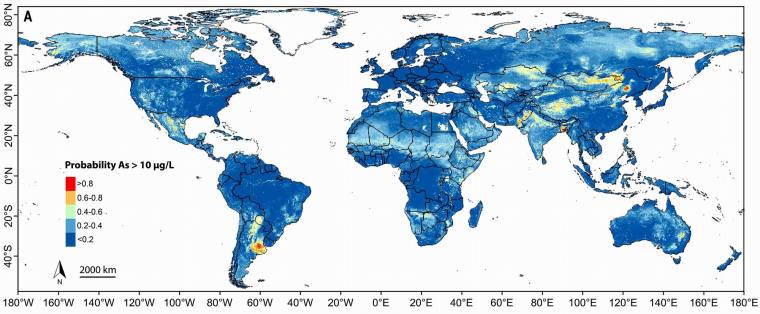
Rather than trying to model the full range of arsenic concentrations, Joel Podgorski and Michael Berg of the Swiss Federal Institute of Aquatic Science and Technology focussed on assessing probabilities that arsenic in well water exceeds the WHO recommended maximum safe level of 10 μg l-1. Their global map highlights areas of concern for environmental health. Thankfully, huge (blue) areas are suggested to present low risk, the pale, yellow, orange and red patches signifying areas of increasing concern. No populated continent is hazard-free. What is very clear is that Asia presents the greatest worries. Most of the Asian ‘hot zones’ are spatially close to large mountain ranges and plateaus. In the case of the Indus and Ganges-Brahmaputra plains the sources for excessive arsenic in groundwater implicated by previous geochemical investigations lie in the Himalaya. The factor common to all major hot spots seems to be rapid transport of huge amounts of sediment released by weathering from areas of high topographic relief, rather than local large-scale mining operations. There are hazardous areas related to historic and active mining, such as the Andes of Bolivia, Peru and Chile and the western USA, but they are tiny by comparison with the dominance of natural arsenic mobilisation.
Despite the WHO recommended maximum of 10 μg l-1 of arsenic, many countries base their policy on levels that are five times higher, largely because of the difficulty of analysing for the lower concentration without expensive analytical facilities. Field analyses are often done using simple semi-quantitative tests based on paper impregnated with reagents that show a colour range for different concentrations, which are unreliable for those lower than 100 μg l-1. Thankfully, despite the many risky areas, most of them have population densities less than 1 per km2.
If you are interested in the geological details of the arsenic problems of Bangladesh, the course text that I produced for the Open University (Drury, S. 2006. Water and well-being: arsenic in Bangladesh. The Open University: Milton Keynes, UK. ISBN 0-7492-1435-X), the course itself (S250 Science in Context) was withdrawn some years ago. It may be possible to arrange a PDF for private use.
See also: Zheng, Y. 2020. Global solutions to a silent poison. Science, v. 368, p. 818-819; DOI: 10.1126/science.abb9746

